




ENGINEERING REDOX ENZYMES FOR BIOCATALYSIS
Prof. Gianfranco Gilardi, Prof. Giovanna Di Nardo,
Prof. Sheila Sadeghi, Dr. Francesca Valetti,
Keywords: protein engineering, oxygenases, bioconversion, bioremediation, drug functionalization, fine chemicals synthesis
The need of green technologies to protect human health and environment are the main inputs for expanding the use of biocatalysis as an alternative to the traditional chemical synthesis methods. Different bacterial oxygenases are available in the research group providing an enzymatic pool that can be used and combined to achieve regio- and stereoselective reactions of biotechnological interest.
Full structural and functional characterization is routinely carried out providing fundamental knowledge in the enzymes properties for the application in the fields of land and water bioremediation, drug late stage functionalization and synthesis of fine chemicals.
In particular, the research is focused on the following enzymes:
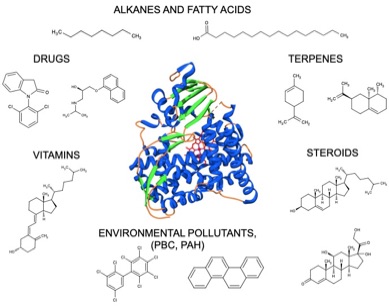
- Cytochromes P450 are known to carry out a wide range of reactions on thousands of substrates. The most common reaction is the hydroxylation of C-H bonds and the activation of unreactive C-H bonds is of particular interest since it is crucial for the biotransformation of compounds especially when they lack functional groups useful for conjugation. Moreover, the C-H bond activation through hydroxylation is considered an important process in the so-called late-stage functionalization that is a challenge for organic chemistry.
- Baeyer-Villiger monooxygenases (BVMOs) are flavin-containing enzymes that have a great value in bioremediation and green chemistry applications. We have cloned and purified a bacterial BVMO enzyme and ongoing projects involve the tuning of the substrate specificity of this enzyme towards compounds of biotechnological importance.
- Catechol 1,2 dioxygenases catalyses the enzymatic cleavage of catechol leading to cis-cis muconic acid that in turn is hydrogenated to produce adipic acid, commonly used for the benzene-free synthesis of nylon-6,6. Mutants of this enzyme have also been generated for the removal of highly toxic chlorinated catechols.